Introduction
Maintaining a robust knowledge database is an essential component of food safety, and the recent cereulide contamination in infant formula provides a critical case study for professionals. A multi-country recall of several infant nutrition products is currently ongoing following the detection of cereulide, which is a toxin produced by the bacterium Bacillus cereus.
The precautionary recalls and withdrawals were initiated in December 2025 and continued into early 2026 as a preventive public health measure. The incident underscores the vulnerabilities in global supply chains and the importance of academic rigor in hazard characterization.
General Information on Bacillus cereus and Cereulide
Bacillus cereus is a widespread, spore-forming bacterium capable of producing two forms of toxin-mediated illness, including an emetic form caused by cereulide. Cereulide is a highly lipophilic and heat-stable dodecadepsipeptide that acts as a potent mitochondrial toxin. The symptoms include sudden nausea, vomiting, and stomach pain within 30 minutes to six hours after ingestion.
The toxin resists heat, acidic conditions, and proteolysis. It is not destroyed by standard infant formula processing or human digestive enzymes. Infants are exceptionally vulnerable because vomiting rapidly disrupts their physiological salt balance. This can lead to severe complications like dehydration. While symptoms are typically self-limiting in adults, neonates and infants under six months face a much higher risk of severe disease.
Root Cause Analysis and Cereulide Contamination Timeline
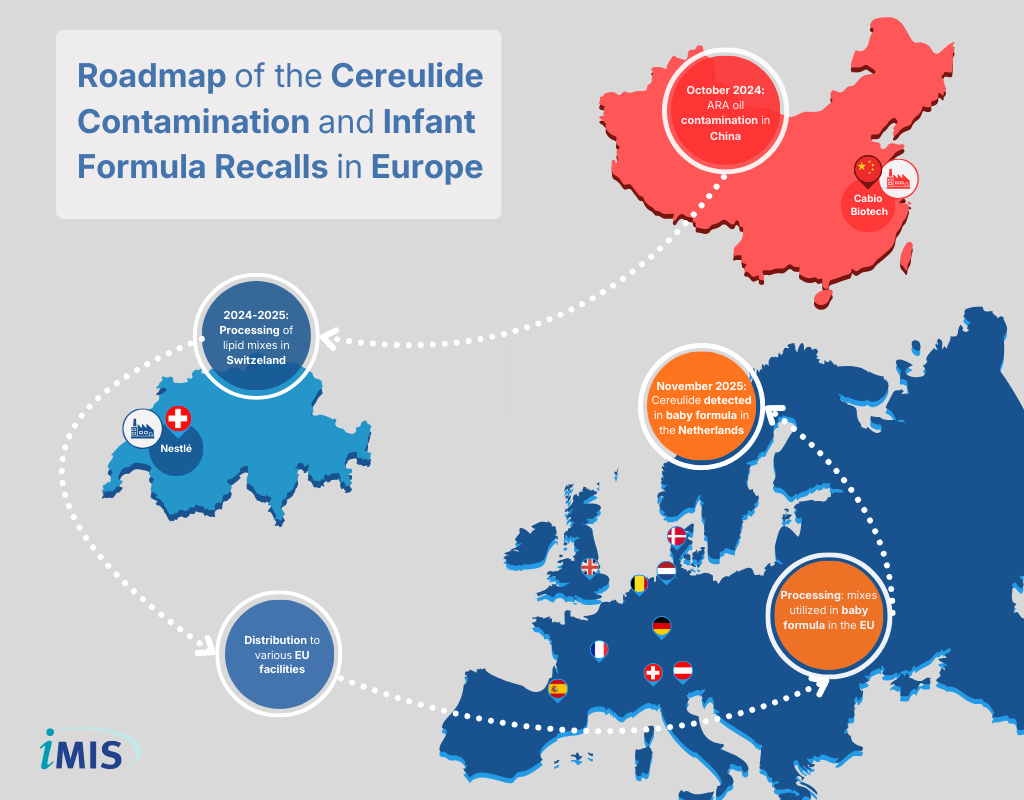
Extensive retrospective testing and international trace-back investigations established a definitive chronological sequence for the cereulide contamination event. Such investigations exposed critical vulnerabilities in global infant formula supply chains, where single-source ingredient contamination cascades into a multinational public health crisis.
- 2024: Contamination of the raw ingredient, specifically arachidonic acid oil (ARA), begins at a primary manufacturing facility in China.
- 2024-2025: The Chinese producer supplies the contaminated raw ingredient to a Swiss manufacturer.
- 2025: The Swiss facility utilizes the contaminated (ARA) to prepare lipid mixes, subsequently distributing the blends to various infant formula manufacturers worldwide.
- July 2025: Deliveries of the contaminated (ARA) oil mixes reach peak cereulide concentrations.
- Late 2025: Formula products incorporating the contaminated lipid blends are manufactured in several European countries, including Germany, Spain, France, the Netherlands, and Switzerland.
- Late November 2025: A manufacturer successfully identifies cereulide in infant formula during testing at a production plant in the Netherlands, triggering initial alerts.
- Early 2026: Clinical cases of gastrointestinal illness in infants begin to emerge and are directly linked to the contaminated formula, prompting widespread recalls across multiple European nations.
Mapping and Distribution
Distribution of the contaminated ARA oil necessitated an international trace-back investigation. The ingredient and resulting oil mixes were shipped globally to manufacturing facilities in countries including Germany, Spain, France, the Netherlands, and Switzerland.
Following distribution of final infant formula products, clinical cases of gastrointestinal illness in infants began to emerge. By early 2026, investigations linked gastrointestinal illnesses and hospitalizations to recalled products across Europe. Austria, Belgium, Denmark, France, Luxembourg, Spain, and the United Kingdom have reported clinical cases associated with the event.
The Impact and Response in the Netherlands
The situation in the Netherlands has been closely monitored by the Dutch Food and Consumer Product Safety Authority (NVWA).
- In mid-December 2025, the NVWA was notified by a manufacturer about a cereulide contamination in a production line of infant formula that was not initially sold in the Netherlands.
The NVWA requested traceability data and shared this information with countries where the products were distributed.
- On 5 January 2026, further investigations revealed that the contamination originated from the raw material arachidonic acid oil, which had been used in products sold on the Dutch market.
The discovery led to the withdrawal and public recall of specific batches of infant formula in the Netherlands, including products from Nestlé “Little Steps 1 and Alfamino” and Danone “Nutrilon”.
- By early February, the NVWA had received 11 reports of children who allegedly became ill after consuming the formula.
Because the source of the contaminated raw material is outside the Netherlands, the NVWA is actively collaborating with other European countries to trace the products and ensure regulatory compliance.
The NVWA has put up a website that answers consumer’s questions: 5 questions about cereulide in infant formula.
EFSA And ECDC Rapid Cereulide Outbreak Assessment
Following widespread detection of cereulide in infant formula, the European Food Safety Authority “EFSA” and the European Centre for Disease Prevention and Control “ECDC” published a joint assessment in February 2026.
- Likelihood of exposure: The joint assessment concludes that current exposure risk is low due to European Union recalls, though additional cases may occur if products remain in households.
- Health impact: Overall impact is low to moderate, but infants under six months are highly vulnerable to severe complications like dehydration, sometimes requiring hospitalization.
- Diagnostic challenges: Confirming cereulide intoxication is difficult because symptoms mimic common viral infections, and routine laboratory testing is not broadly available.
Cereulide Preventative Measures: Industry-Wide Vigilance
Preventing future occurrences requires stringent raw material testing and adherence to updated safety thresholds. While recent incidents heavily spotlight vulnerabilities within infant nutrition, the broader food sector must remember that cereulide contamination remains a pervasive threat across various matrices. The potent toxin frequently affects farinaceous foods, cooked rice, pasta dishes, and ready-to-eat meals subjected to improper temperature control.
To protect consumer health and maintain brand integrity, producers across all sectors must implement rigorous control measures:
- The EFSA recently established an acute reference dose “ARfD” of 0.014 µg/kg body weight for cereulide in infants.
- Complying with the newly defined toxicological threshold means cereulide concentrations must not exceed 0.054 µg/L in reconstituted infant formula and 0.1 µg/L in follow-on formula.
- Manufacturers must implement mandatory testing of all arachidonic acid oil deliveries using validated analytical methods, such as liquid chromatography tandem mass spectrometry (LC-MS/MS).
- When analyzing powdered products, a prior hydration step is absolutely essential; direct extraction yields from dry matrices are significantly low and produce dangerous false-negative results.
- Industry quality assurance programs must establish enhanced supplier verification protocols for all lipid-based ingredients to prevent single-source failures.
Navigating complex toxicological thresholds and implementing validated testing methodologies requires specialized technical expertise. Facilities struggling to map supply chain risks or validate protocols should seek expert food safety consultancy.
Professional guidance ensures that internal hazard control plans remain resilient against emerging microbiological threats before a critical incident occurs.
Automated Hazard Management with iMIS Food
Managing microbiological risks requires dynamic digital tools. iMIS Food simplifies complex compliance through an integrated, up-to-date database.
- Access comprehensive bacterial and viral hazard tables, now also fully translated into Dutch for seamless use by local quality teams.
- Unlock highly specific data on emerging hazards like cereulide, including actionable prevention strategies, by utilizing the advanced compliance module.
- Incorporate specialized prevention protocols directly into an active HACCP study automatically, saving time and reducing manual errors.
- Receive automatic system updates during global contamination events, ensuring that manufacturing facilities remain completely prepared and internal safety frameworks stay resilient.
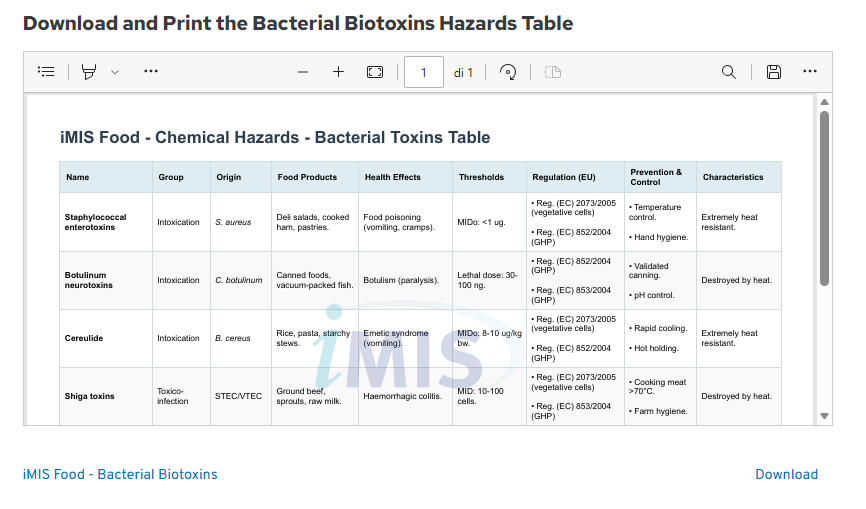
New: Bacterial Biotoxins Table for Enhanced Prevention
The recent cereulide incident has exposed significant gaps in traditional supply chain monitoring, where reliance on raw material suppliers failed to prevent a multinational crisis. By integrating the new Bacterial Biotoxins overview into your HACCP plan, you can avoid these pitfalls.
This tool centralizes critical data on potent substances like cereulide, which are often heat-stable and survive standard processing. Having this information readily available allows for immediate, proactive adjustment of monitoring procedures and cold chain management, preventing hazardous byproducts from ever entering the finished product.
More Reference Websites For Further Information
For ongoing updates and regulatory guidance, professionals should consult the following platforms:
- The EFSA website provides scientific risk assessments and updated toxicological thresholds.
- The ECDC platform offers epidemiological updates and outbreak assessments.
- The EU Rapid Alert System for Food and Feed (RASFF) portal is essential for tracking real-time information on product recalls and border rejections.
- The NVWA website publishes national safety warnings and updates regarding recall status in the Netherlands.
Sources
- European Centre for Disease Prevention and Control. (2026, February). Lower risk of exposure to cereulide following continued recall of infant formula products across Europe. https://www.ecdc.europa.eu/en/news-events/lower-risk-exposure-cereulide-following-continued-recall-infant-formula-products-across
- European Food Safety Authority. (2026). EFSA provides rapid risk assessment on cereulide in infant formula. https://www.efsa.europa.eu/en/news/efsa-provides-rapid-risk-assessment-cereulide-infant-formula
- European Food Safety Authority. (2026). Precautionary global recall of infant nutrition products following detection of Bacillus cereus. https://www.efsa.europa.eu/en/news/precautionary-global-recall-infant-nutrition-products-following-detection-bacillus-cereus
- European Food Safety Authority. (2026). Scientific risk assessment of cereulide in infant formula. EFSA Journal, 24(2), 9941. https://efsa.onlinelibrary.wiley.com/doi/abs/10.2903/j.efsa.2026.9941
- Nederlandse Voedsel- en Warenautoriteit. (2026). 5 vragen over cereulide in zuigelingenvoeding. https://www.nvwa.nl/actueel/actuele-onderwerpen/5-vragen-over-cereulide-in-zuigelingenvoeding
- Nederlandse Voedsel- en Warenautoriteit. (2026, January 23). NVWA monitort situatie zuigelingenvoeding nauwgezet. https://www.nvwa.nl/actueel/nieuws/2026/01/23/nvwa-monitort-situatie-zuigelingenvoeding-nauwgezet
Related articles to Tracing the Infant Formula Cereulide Outbreak: From Chinese Raw Materials to European Recalls
Many customers and visitors to this page 'Tracing the Infant Formula Cereulide Outbreak: From Chinese Raw Materials to European Recalls' also viewed the articles and manuals listed below:
